
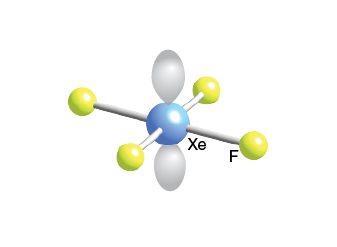
The Lewis structure of XeOF4 is as follows: However, in XeOF4, it forms bonds with oxygen and fluorine to complete its octet. Xenon, being a noble gas, already has a stable electron configuration. To determine the Lewis structure, we follow the octet rule, which states that atoms tend to gain, lose, or share electrons to achieve a stable electron configuration with 8 valence electrons. Xenon (Xe) has 8 valence electrons, while oxygen (O) and fluorine (F) have 6 and 7 valence electrons, respectively. In the Lewis dot structure of XeOF4, we represent the valence electrons of each atom as dots around their respective symbols. Its Lewis structure helps us understand the arrangement of these atoms and the bonding within the molecule. XeOF4, also known as xenon oxytetrafluoride, is a chemical compound composed of xenon, oxygen, and fluorine atoms. Lewis structure Detailed Explanation of XeOF4 In the following sections, we will delve deeper into the concepts and theories that govern the structure and properties of this compound. These key terms provide a solid foundation for understanding the intricacies of XeOF4 molecular geometry. XeOF4 resonance structures: Different Lewis dot structures that can be drawn for XeOF4, highlighting the delocalization of electrons. XeOF4 valence electrons: The electrons in the outermost energy level of the xenon atom that participate in chemical bonding. XeOF4 molecular shape: The three-dimensional arrangement of atoms in the XeOF4 molecule, which is square pyramidal.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
